Helping Payers Thrive in a Changing Healthcare System
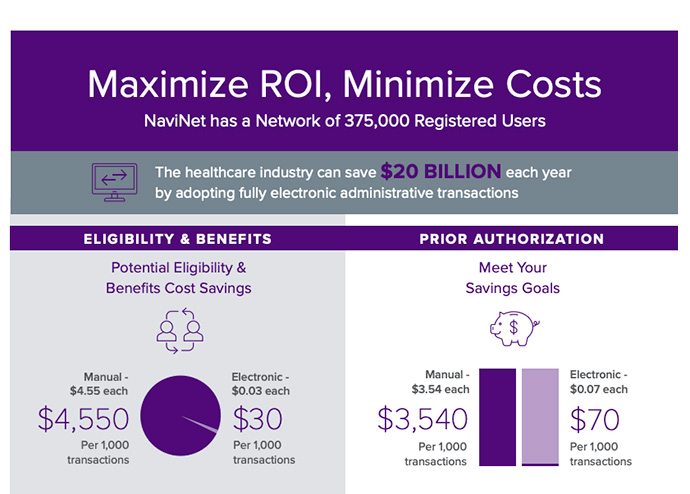
Estimated amount the healthcare industry could save annually with electronic transactions.1
Direct cancer-related healthcare spending in the U.S.2
Percentage of healthcare spending in the U.S. attributed to waste.3
Health plans are under immense pressure to ensure timely, quality and cost-effective care across their membership. To achieve this goal, payers must work collaboratively with their provider networks using technology that simplifies workflows, empowers confident decisions, improves care coordination and boosts efficiencies.

Payer-Provider Collaboration
NaviNet® suite of solutions enables payers and providers to share vital administrative and clinical information in near real-time through the NaviNet portal.
- Enable bi-directional information exchange with your provider network
- Increase operational efficiencies by automating manual processes
- Eliminate redundancy and significantly save on operational costs by shifting to electronic authorizations
Treatment Plan Validation + Decision Support
Eviti Connect® helps payers better manage costs for oncology and autoimmune conditions through a streamlined authorization workflow that facilitates expedited care for members.
- Reduce care costs for oncology and autoimmune conditions
- Eliminate inappropriate member care
- Support value-based care for your members